Abstract
Introduction: Development of massive sequencing of whole exomes (WES; whole exome sequencing) has revolutionized DNA sequencing, leading to several studies detailing the genetic profile of patients with several types of neoplasms, including MPNs and MDS/MPNs. One important challenge is to identify recurrently mutated genes that may play a role in disease pathogenesis. It is important to consider that cohort size is critical in the analysis to determine putative oncogenic driver genes, and most cohorts published in MPNs and MDS/MPNs have a limited sample size. One possibility to circumvent this limitations is to combine datasets of different studies. Herein we report the combined analysis of WES data of 403 patients with these disorders, including 124 patients from our center and 279 cases that were previously reported in the literature.
Patients and Methods:
We analyzed a combined cohort of 403 patients in this study with a diagnosis of Ph-negative MPN or MDS/MPN. For the 124 patients included from our center, reads were aligned and processed following Genome Analysis Toolkit best practices, and somatic variants were called combining the output of SomaticSniper, Mutect and Pindel. Regarding public data, we selected 6 studies that evaluated patients with MPNs and MDS/MPNs through WES and where sample-level data on all individual mutations that were identified were available. Data on identified mutations was extracted from supplementary material of the articles. Combined data on genomic mutations from the whole cohort was used to predict driver genes using softwares MutSigCV and IntOGen (www.intogen.org). Genes considered as being significantly mutated were genes with q-value <0.1 and/or genes with a mutational hotspot.
Results:
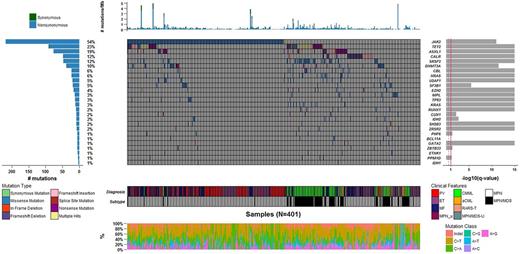
Diagnosis of the cohort included essential thrombocythemia (ET; N=110), polycythemia vera (PV; N=93), myelofibrosis (MF; N=98), chronic myelomonocytic leukemia (CMML; N=82), atypical CML (aCML; N=15) and more rare disorders (MPN-unclassified [N=2], MDS/MPN-unclassified [N=2] and refractory anemia with ring sideroblasts and thrombocytosis [N=1]). There were a total of 7519 identified mutations, with a median of 9 nonsilent coding mutations per sample (range 0-311 mutations). There were 2 patients (both with a diagnosis of ET) who had no somatic mutations identifed. Analysis of the mutational data using both MutSigCV and IntOGen algorithms revealed a total of 26 genes considered as putative driver genes in these neoplasms: JAK2, TET2, ASXL1, SRSF2, CALR, DNMT3A, SF3B1, CBL, NRAS, EZH2, U2AF1, RUNX1, KRAS, CUX1, MPL, ZRSR2, TP53, IDH2, PHF6, SH2B3, BCL11A, GATA2, ZBTB33, ETNK1, IDH1 and PPM1D (Figure) . A mutation in at least one these genes was found in 92.5% of samples. Among these 26 genes, 2 genes were not previously reported to be mutated in these diseases (ZBTB33 and BCL11A), and one gene was previously reported to be associated with other types of neoplastic diseases (PPM1D). ZBTB33 encodes the transcriptional regulator Kaiso and was found to be mutated in 6 samples (MF=2, ET=2, PV=1, CMML=1). Three mutations were frameshift indels, and three mutations were missense mutations, one of them (p.I37T) located in the POZ/BTB domain that regulates protein homodimerization. The second gene that was found to be recurrently mutated is BCL11A, which encodes a zinc-finger protein. There were 6 cases of BCL11A mutations, most of them in MDS/MPNs (CMML=3, aCML=2, MF=1). Mutations were all missense and clustered in two asparagine residues located at positions 391 and 756. Finally, four patients (CMML=2, PV=1, MF=1) presented with truncating mutations in the last exon of the PPM1D gene. These mutations have been reported to be activating, leading to an increase in activity of the Wip1 phosphatase and suppression of p53 activity. PPM1D mutations have been reported to be associated with clonal hematopoiesis, MDS and solid tumors. To the best of our knowledge this is one of the first reports detailing their presence in MPNs.
Conclusions: Combining datasets across different studies has the potential to increase power and saturate discovery of recurrently mutated genes. This permits prioritization of genes for functional analysis to confirm their role in disease pathogenesis. Larger cohorts than the ones analyzed here may provide even more meaningful data on putative driver genes in MPNs and MDS/MPNs.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.